Oxyphencyclimine
Chemical compound
- A03AA01 (WHO)
- (1-Methyl-1,4,5,6-tetrahydropyrimidin-2-yl)methyl 2-cyclohexyl-2-hydroxy-2-phenylacetate
- 125-53-1
 Y
Y
- 4642
- 7256
- 4481
 N
N
- 4V44H1O8XI
- D08325
 Y
Y
- ChEMBL1200891
 N
N
- DTXSID4023410

InChI
- InChI=1S/C20H28N2O3/c1-22-14-8-13-21-18(22)15-25-19(23)20(24,16-9-4-2-5-10-16)17-11-6-3-7-12-17/h2,4-5,9-10,17,24H,3,6-8,11-15H2,1H3
 N
N - Key:DUDKAZCAISNGQN-UHFFFAOYSA-N
 N
N
 N
N Y (what is this?) (verify)
Y (what is this?) (verify)Oxyphencyclimine is a muscarinic receptor antagonist, given orally to treat peptic ulcer disease and gastrointestinal spasms. It has antispasmodic and antimotility properties.
Synthesis
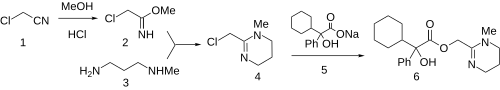
The reaction of chloroacetonitrile (1) with methanol and hydrogen chloride leads to the corresponding iminoether (Pinner reaction). Condensation of 2 with 3-methylaminopropylamine gives (3) gives the corresponding tetrahydropyrimidine (4). Displacement of the halogen with the sodium salt 5 affords oxyphencyclimine (6).
References
- ^ Faust JA, Mori A, Sahyun M (1959). "Antispasmodics: Esters of Heterocyclic Alcohols". Journal of the American Chemical Society. 81 (9): 2214. doi:10.1021/ja01518a051.
- ^ GB 795758, "Amino esters and the preparation thereof", published 1958-05-28, assigned to Charles Pfizer & Co. Inc.
- v
- t
- e
Drugs for functional gastrointestinal disorders (A03)
functional
bowel
disorders
| Antimuscarinics |
| ||||
|---|---|---|---|---|---|
| Phosphodiesterase inhibitors | |||||
| Acting on serotonin receptors |
| ||||
| Other |
and derivatives
(antimuscarinics)
- Tertiary amines: Atropine
- Hyoscyamine
- Quaternary ammonium compounds:
- Cimetropium bromide
- Fentonium bromide
- Methylatropine
- Primarily dopamine antagonists
- 5-HT4 agonists
- Acotiamide
- Ilebopride
- Mosapride
 | This drug article relating to the gastrointestinal system is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e










